
Polish scientists develop ‘invisible’ electrode that could boost lasers, sensors and thermal imaging
Polish researchers have developed a new type of electrode that combines high electrical conductivity with unusually high transparency to infrared radiation, a combination that could improve the performance of infrared detectors, lasers, thermal cameras and other optoelectronic devices.
-

CMS experiment at CERN finds W boson mass consistent with standard model
The CMS experiment at CERN has measured the mass of the W boson with a precision approaching 10 MeV, producing a result consistent with the standard model of particle physics and challenging earlier findings that hinted at possible new physics.
-

Warsaw scientists develop AI-assisted imaging method for semiconductor nanostructures - thanks to black holes
Scientists from the Warsaw University of Technology have developed a computational imaging method that can precisely measure semiconductor nanostructures from a single optical image, potentially speeding up quality control in photonic chip manufacturing and quantum technologies.
-

Polish-Chinese team prints ‘vortex beam generator’ directly on optical fibre
Researchers from Poland and China have developed a method for printing microscopic structures directly onto the end of optical fibres to generate so-called vortex beams, a technology seen as a potential route toward higher-capacity optical communication systems.
-
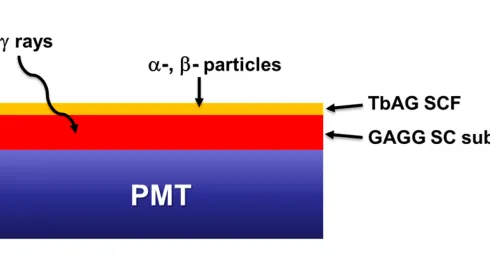
Polish-Ukrainian team develops detector to identify multiple radiation types simultaneously
Scientists from Poland and Ukraine have developed a new type of detector capable of simultaneously identifying different kinds of radiation, a breakthrough that could improve medical treatments and radiation protection systems.
-

AI model predicts atomic motion in molecular simulations, boosting speed nearly 100-fold
An international team of researchers has developed an artificial intelligence model that can predict the motion of atoms in molecular systems directly, dramatically speeding up simulations used in chemistry, physics, and materials science. The model, called MDtrajNet, bypasses the traditional step-by-step calculations of atomic positions, a process that can require millions of steps and substantial computing power.
-

Polish scientists develop quantum dot system to deliver chemotherapy drugs directly to tumours
Scientists from the Warsaw University of Technology are developing a system designed to deliver chemotherapy drugs directly to tumours using nanometric carriers, which could reduce damage to healthy cells and improve treatment effectiveness.
-
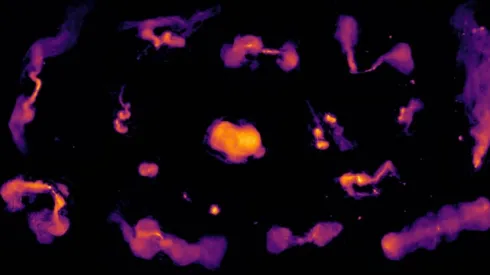
Scientists create largest and most detailed radio map of the Universe
An international team of scientists has produced the largest and most detailed radio map of the Universe to date, revealing 13.7 million active galaxies and pinpointing the locations of supermassive black holes.
-

Poland emerges as key player in CERN’s ALICE experiment after Russia’s exit, says Polish physicist
Poland’s role in the ALICE experiment at European Organization for Nuclear Research (CERN) has grown significantly following the end of cooperation with Russia, according to Krystian Rosłon of the Warsaw University of Technology.
-

Polish scientists develop nanocomposite that produces biocides on demand
Polish scientists have developed a silica-based nanocomposite known as B-STING, that can automatically generate biocidal substances in response to microorganisms, effectively targeting bacteria, fungi, and viruses while remaining safe for human cells.















